- India Medical Mission 2018 - November 1, 2018
- Sleep Disorders: Sleep Apnea and Upper Stimulation Therapy - August 25, 2015
- The Naked Vocalist Podcast Featuring Dr. Reena Gupta - May 27, 2015
- New Therapy for Sleep Apnea – First Sleep Pacemaker placed in California at Osborne Head and Neck Institute. - December 12, 2014
- Boxer’s Ear: Can your ear explode? - December 12, 2014
- Nose Picking (Rhinotillexis) and Septal Perforations: Why I should stop picking my nose…? - November 24, 2014
- Deviated Septum and Septal Perforation - July 28, 2014
- Hereditary Hemorrhagic Telangiectasia: Nasal Septal Perforation Repair - June 25, 2014
- Dr. Mantle recognized at the Beverly Hills Medical Science Academy Awards - May 8, 2014
- Commonly Misdiagnosed Pathologies: Arteriovenous Malformations - April 9, 2014
The first sleep pacemaker in California and the Western United States was implanted at Osborne Head and Neck Institute, Los Angeles, California, on November 17, 2014. The patient has had a remarkable recovery and improvement in symptoms.
Obstructive Sleep Apnea (OSA) is a serious condition, where the body receives inadequate oxygenation during sleep. Muscular collapse of the airway during sleep results in obstruction and cessation of breathing. During the period of time when breathing is halted, blood oxygen levels decrease. This is referred to as apnea. The body reacts to apneic events by rousing itself from sleep and disturbing the normal sleep cycle. Sleep apnea can often lead to excessive daytime sleepiness, difficulty concentrating, distractibility, mood changes, and memory problems in the short term. Long-term effects of untreated OSA are associated with high blood pressure, cardiac disease, and stroke.
Historically, OSA has been extremely difficult to treat. Despite knowledge of the risks of untreated sleep apnea, patients often elect to forego treatment. This is usually because treatment options are suboptimal and can include uncomfortable masks, pressurized breathing machines (CPAP), or painful surgery.
The FDA has recently approved a new, fully-implantable device for the treatment of obstructive sleep apnea. The most successful device on the market is made by Inspire Medical Systems, and consists of a pacemaker-type generator that stimulates key airway muscles to prevent obstruction during sleep. This upper airway stimulation (UAS) allows for continuous oxygenation during sleep and resolution of apneas.
The device has shown remarkable success, with all implanted patients exhibiting significant improvement in measurements related to sleep apnea. During a simple, outpatient procedure, a thin stimulating wire is wrapped around the nerve responsible contraction of the tongue muscles. This wire is then connected to a pulse generator in the upper chest and a breathing sensor near the rib muscles. By keeping airway muscles open, patients are able to discontinue use of continuous airway pressure machines (CPAP). Risks of OSA-related diseases, such as stroke, cardiac arrhythmias, and hypertension are significantly decreased with successful treatment. Further, snoring resolves in most patients, resulting in an improved quality of sleep for the patient and their bed partner.
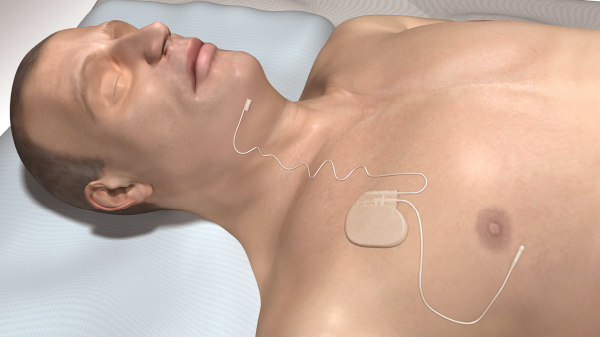
Figure: Depiction of a patient with an upper airway stimulation implant. UAS therapy consists of a small generator that is placed in the chest much like a pacemaker. A thin connection is made from the generator to key airway muscle nerves. An additional connection is made to muscles along the rib cage to monitor respiration.
The first sleep pacemaker in the Western United States was implanted at Osborne Head and Neck Institute on November 17th, 2014. Followed by the second on November 18th, 2014. The patients have both gone on to have remarkable improvements after their uncomplicated surgery.
“Sleep apnea as a disease is difficult, with the significant health problems that accompany it. What has made it even worse are the treatment options, which have been marginally effective at best and ineffective and intolerable at worst. With this new device, we have addressed most of the major issues surrounding the care of sleep apnea. There is now an effective and well-tolerated option for patients who struggle with sleep apnea.”
– Dr. Ryan Osborne, Director of the Division of Head and Neck Surgery at Osborne and Neck Institute.
To learn more about Dr. Ryan Osborne or obstructive sleep apnea evaluation and treatment, please visit http://ohnisleepapneatreatment.com/.